For many people in developing countries, the struggle for access to health care comes with a risk. Those patients can fall victim to falsified medications that fail to treat their conditions, with the potential to cause harm and, in some cases, death.
Now, a new study will take aim at illicit supply chain operations to improve detection of falsified medications before they harm patients. The project is a collaboration between the University of Notre Dame and Northwestern University.
The study is one of nine projects awarded funding through the National Science Foundation’s Early-Concept Grants for Exploratory Research (EAGER) focusing on disrupting illicit supply chain networks.
“Many countries in the developing world do not have the infrastructure or resources to keep low-quality medicines off market shelves,” said Marya Lieberman, professor in the Department of Chemistry and Biochemistry at the University of Notre Dame, who is leading the study with Karen Smilowitz at Northwestern University. “Through this project, we will work to detect falsified medicines efficiently in low-resource settings, and track the supply chains through which these bad-quality products move. By working with partners in the health system and with regulatory agencies, we hope to protect patients from dangerous fake drugs.”
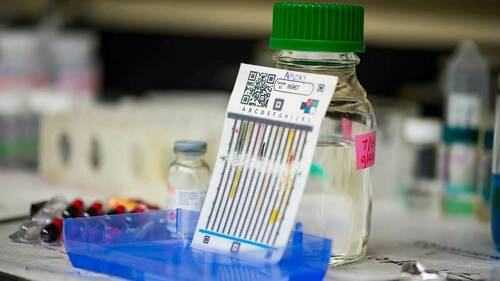
Lieberman notably developed a cost-effective and efficient method of finding falsified medications in 2013 using paper analytical devices (PADs). PADs are paper cards containing a set of chemical color tests that identify suspicious medications by showing whether a medicine contains the necessary active ingredients.
The new two-year study will employ covert shoppers to collect 1,200 samples of essential medications in Kenya, Malawi and Bangladesh. The team will use PADs and chemical analysis to detect fake or low-quality drugs, and report results to regulatory agencies and the World Health Organization’s Rapid Alert system.
“The PADs give us additional chemical information that can help to trace how fake medicines are manufactured,” Lieberman said. “We want to see how many producers are active in the supply chains for these illegal products and learn about weak points in these supply chains. There are a lot of similarities between the supply chains for bad-quality medicines and the supply chains for illicit drugs.”
If the project succeeds, Lieberman sees the effort someday helping with issues in the U.S. “As we develop methods for detecting and eliminating bad-quality medicines,” she said, “we will also test them to see if they could help control the opioid overdose epidemic in the United States.”
Contact: Jessica Sieff, assistant director of media relations, 574-631-3933, jsieff@nd.edu