U.S. Department of Energy (DOE) Secretary Samuel W. Bodman has announced that a University of Notre Dame project led by College of Engineering faculty members Edward J. Maginn, Joan F. Brennecke, and William Schneider, has been awarded a grant in excess of $3 million to explore the use of ionic liquids as solvents for the post-combustion capture of carbon dioxide (CO 2 ).
The project was one of nine proposals awarded a total of nearly $24 million through DOEs new carbon sequestration program.
The purpose of the DOE program is to encourage the development of novel and cost-effective technologies for the capture and sequestration of CO 2 produced in coal-fired power plants. It supports President Bushs Global Climate Change Initiative, which calls for an 18 percent reduction inU.S.greenhouse gas intensitythe ratio of emissions to economic outputby 2012.
Carbon (CO 2 ) sequestration promises to significantly reduceAmericas greenhouse gas emissions even as our economy grows,Bodman said.This combination helps protect the global climate, while promoting job creation and a high standard of living.
According to Bodman, the key to successful carbon sequestration is developing appropriate technologies to capture greenhouse gases before they are released into the atmosphere.
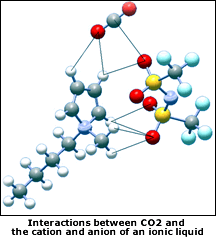
The goal of the Notre Dame project is to exploit the unique properties of ionic liquids so they can be used as solvents for the cost-effective capture of CO 2 .
Ionic liquids are salts that are liquid in their pure state, near ambient conditions,Maginn said.They have a number of interesting and useful properties, including high thermal stability and very low volatility. We have been working with them for some time and have discovered that these properties make them suitable for a wide range of technical applications.
Two years ago, as part of a project sponsored by the DOEs National Energy Technology Laboratory, a research team led by Maginn and Brennecke demonstrated that ionic liquids have the potential to efficiently capture CO 2 from the flue gas of coal-fired power plants. Ionic liquids, they believe, are potentially a pivotal component in an integrated system that can safely and economically sequester combustion-generated CO 2 , thereby mitigating its impact on climate change.
The Notre Dame team will be developing ionic liquids specifically designed for this task. To accomplish this, they are using a combination of atomistic simulations and targeted experimentation. Working with Babcock and Wilcox,Barberton,Ohio; Merck KGaA,Darmstadt,Germany; DTE Energy,Detroit; and Trimeric Corporation,Buda,Texas, the research group will conduct design and economic studies of various processes that use ionic liquids. They also will build a working prototype CO 2 capture unit.
Brennecke is the director of the Notre Dame Energy Center and the Keating-Crawford Professor of Chemical and Biomolecular Engineering. Maginn is a professor and Schneider is an associate professor of chemical and biomolecular engineering.
More information about Notre Dames work with ionic liquids and other energy related research can be found at: http://energycenter.nd.edu .
TopicID: 20402